Liposomes have been widely used as model membranes and in drug delivery, as their basic properties can easily be varied to target desired properties (lipid composition, concentration, water content, media (different buffers, varying ionic strength, pH, etc.) as well as lamellarity (unilamellar, large or small (Large Unilamellar Vesicles, LUVs, and Small Unilamellar Vesicles, SUVs), and multilamellar vesicles, MLVs)). In these studies, one must characterize the pure liposomes as well as their mixtures with other molecules (proteins, peptides, drugs), to ascertain the influence of the added component in the liposome phase behavior.
DSC is a fundamental technique for the characterization of membrane behavior, providing all thermodynamic parameters for temperature-induced transitions. Furthermore, for mixtures of membranes with other components (drugs, proteins, peptides, polymers, etc.) DSC represents a first-line screening method, providing information on the effect(s) of the added components on membrane behavior.
Lipids are fundamental constituents of cell membranes, forming a lipid bilayer into or onto the surface of which proteins and other constituents are incorporated or bound[1-4]. Biological membranes require a mainly fluid environment for proper function, but different states of fluidity are also important, as these provide membrane compartmentalization (such as lipid-raft domains in plasma membranes, where a liquid-ordered phase is present[5,6]) related to protein insertion and function. Consequently, there is great interest in the study and detailed characterization of lipid bilayer membranes[7] for a deeper understanding of their biological function[8].
When lipids are brought into contact with water, the lyotropic phase either forms spontaneously at room temperature or the suspension must be sonicated or vortexed at higher temperature, above its transition temperature into the liquid-crystalline phase. It was recognized early on that changes in concentration and/or temperature can lead to different phases, a process called lyotropic and thermotropic mesomorphism[1]. Phospholipids then form lyotropic lamellar phases in the form of either multilamellar, small oligolamellar, or unilamellar vesicles. Changes in temperature can then lead to temperature-induced transitions between different types of lamellar phase, or to further lyotropic phases, such as inverted hexagonal or bicontinuous cubic phases[7].
Liposomes have been widely used as model membranes and in drug delivery[6,9-19], as their basic properties can easily be varied to target desired properties (lipid composition, concentration, water content, media (different buffers, varying ionic strength, pH, etc.) as well as lamellarity (unilamellar, large or small (Large Unilamellar Vesicles, LUVs, and Small Unilamellar Vesicles, SUVs), and multilamellar vesicles, MLVs)). In these studies, one must characterize the pure liposomes as well as their mixtures with other molecules (proteins, peptides, drugs), to ascertain the influence of the added component in the liposome phase behavior[6,20-26].
The ideal experimental technique for the determination of thermotropic phase transitions in lyotropic lipid phases for membranes composed of pure lipids, as well as their mixtures with any other component, is Differential Scanning Calorimetry (DSC)[24,27]. Consequently, this technique has been widely used for this purpose, and significant data has been made available in the literature[13,28-31], including some review papers[6,32-35].
Most studies focus only on transition temperature(s), the property which it is easiest to determine with high reproducibility. Nevertheless, with an appropriate preparation protocol, transition enthalpies can also be obtained by current DSC instruments, with good precision and reproducibility for transitions with high enthalpy (lamellar phase transitions). In most lipids, the main transition is from gel (Lb') to liquid crystalline phase (La), a fast and highly reversible transition which is characterized by the co‑operative melting of the hydrocarbon chains and a high enthalpy DSC peak. In phosphatidylcholines the so-called pre-transition from planar gel (Lb') to rippled phase (Pb’) has a low enthalpy and is very sensitive to sample preparation and to the presence of impurities. Even so, it can also be easily obtained in many cases, but it should be stressed that it is more sensitive to the scan rate, with lower scan rates resulting in lower transition temperatures (Figure 1).
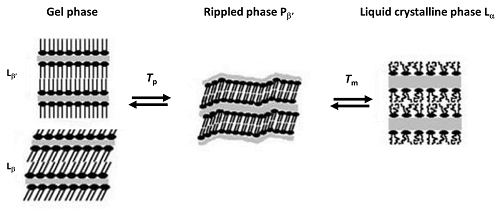
Figure 1. Pre (Tp) and main (Tm) transitions in liposomes of phosphatidylcholines (adapted from Koynova and Caffrey, 1998b[31])
The transition from liquid crystalline phase (La) to hexagonal phase (H), common in phosphatidylethanolamines, has a much lower enthalpy, but at high enough concentrations it can also be measured by high sensitivity DSC instruments. There are additional transitions which are observed under certain experimental conditions, but these will not be covered within this application note.
When liposomes are used as model membranes, we aim to observe the effect of added components on liposome behavior, to reveal fundamental aspects of their mechanism of action. On the other hand, if liposomes are used as drug carriers, the changes in their behavior upon drug loading, and dependence on the method of drug incorporation, pH or other variables, all have to be determined and characterized. In all these studies, DSC is a first line method which provides reliable and comprehensive information on the bulk action.
DMPC (1,2-dimyristoyl-sn-glycero-3-phosphocholine) was used for this extensive methodological study. Complementary data is also provided for mixtures of DMPC and DMPG (1,2-dimyristoyl-sn-glycero-3-[phosphorac-(1-glycerol)]) in a 3:1 molar mixture - this was chosen as an example of a lipid mixture with close-to-ideal miscibility and behavior, and POPE and POPG (1-palmitoyl-2-oleoyl-sn-glycero-3-phosphoethanolamine and 1-palmitoyl-2-oleoyl-sn-glycero-3-phospho-(1'-rac-glycerol), respectively), also in a 3:1 molar mixture, as an example of a lipid mixture with pronounced non-ideal behavior.
In order to achieve reproducible results, a strict preparation protocol must be followed. Preparation details vary among users, but the main steps are as follows:
In most preparation methods, liposomes are initially obtained as MLVs. The most commonly-used preparation method is the lipid film method. However, liposomes comprised of a single lipid can be obtained by direct hydration.
When MLVs are to be used, they should be kept in the refrigerator (4°C) for at least 8 hours prior to DSC analysis.
When MLVs are to be used, they should be kept in the refrigerator (4°C) for at least 8 hours prior to DSC analysis.
In most cases, LUVs are the best form of liposome to use, in order to ensure high reproducibility of results. MLVs sediment over time, so extreme care must be taken when pipetting a sample to be analyzed; SUVs become unstable over time, as due to their high degree of curvature they tend to fuse into larger vesicles.
LUVs are prepared from the MLVs by extrusion. The extruder contains a thermostated cell with a recirculating water bath that keeps it at a temperature ~10°C above the gel to liquid crystalline transition temperature. The liposome suspension is then passed through filters of decreasing pore size (600 nm, 200 nm and 100 nm; 5, 10 and 10 times, respectively), under inert (N2) atmosphere.
After preparation, the lipid samples are stored overnight in the refrigerator at 4°C before being used for DSC analysis.
Sample characterization is completed by measuring the diameter of the vesicles using Dynamic Light Scattering. Several measurements are performed, and the average result taken, reported together with its uncertainty (e.g., 2x standard deviation of the mean, or any other dispersion parameter that should be stated). An example result is shown below in Figure 2, where a good correlogram was obtained, with a polydispersity index of 0.1 and a single peak, leading to a measured size (based on intensity) of 146 nm ± 8 nm.
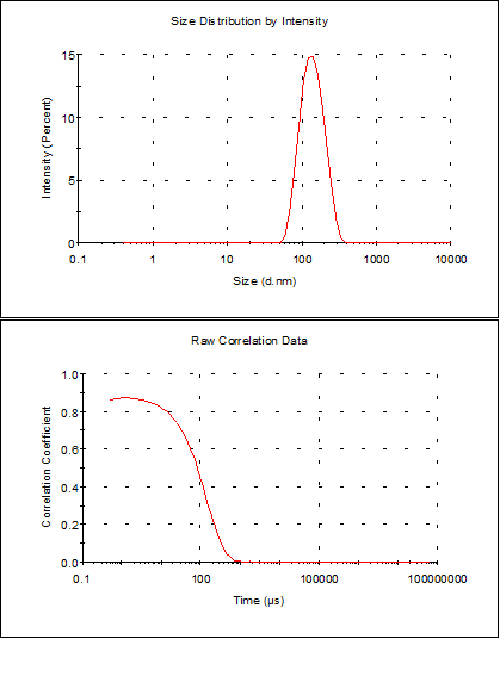
Figure 2. Results obtained by DLS for a liposome preparation of DMPC/DMPG 3:1 molar mixture in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4) after extrusion
The accurate phospholipid concentration is then determined by the phosphomolybdate method[36]. This is fundamental for calculating the accurate enthalpy, as some lipid (typically 3-5%) is lost in the extrusion process, and the accurate concentration is required for the correct enthalpy of transition to be reported.
In all cases, the VP-DSC was loaded with degassed buffer on both sides on the evening prior to the experiments, and the instrument was left to scan overnight using the selected scanning program (initial and final temperature, waiting time before and after the scan, gain, filter). The sample, previously degassed at ~2°C below the starting temperature, is then loaded into the sample compartment of the calorimeter without stopping the acquisition program (loading “in cycle”),
The advised gain to be used for liposome transitions is “high”, as they occur over a very narrow temperature interval. Indeed, in some cases it can be useful to test different gains, and use the most appropriate one. Therefore, all the experiments were performed with a high gain selected (excepting the set of experiments where the gain was tested).
After the experiment, data analysis involved:
It is usually observed that even for liposomes of a single lipid, the results obtained from the first scan are slightly different from the following ones, especially regarding the pre-transition in MLVs. Therefore, one should always use the values of the second or later scans, and report which scan the values refer to. In this work with DMPC and also with the lipid mixtures, the reported values are always calculated from the second scan.
Cleaning is fundamental in any experimental measurement to ensure obtaining reliable data. The cleaning procedure to adopt depends on the lipid used, the length of measurement, and the existence of precipitation/aggregation. In most measurements where no precipitation/aggregation occurs, and no extensive repeated runs are made using the same sample (i.e. no more than 3 or 4 replicates), after removing the sample the cell should be washed manually with water at least 15 times. The cell should then be flushed with buffer prior to fresh sample loading.
If precipitation/aggregation occurs, the sample compartment must be washed either with detergent or with nitric acid solution. The cleaning agent is loaded and the temperature can be increased if desired to improve cleaning. After ~10 min, the sample compartment is removed, extensively washed manually (> 15 times), and then washed automatically with around 250 cm3 water in continuous flow.
After a complete set of measurements - typically one week for lipids - a complete washing procedure should be performed and the system loaded with water and left running baseline scans overnight to ascertain correct cleaning.
Taking DMPC as a single lipid model, DSC results are provided for various experimental conditions:
In all cases except 4 (varying gain), the values reported here were obtained under high gain.
MLVs and LUVs from DMPC were prepared in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4) as described above.
DSC traces can be seen in Figure 3 for the direct hydration (A) and lipid film (B) preparation methods.

Figure 3. DSC traces of MLVs from DMPC in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4) for the direct hydration (A) and lipid film (B) preparation methods. The curves correspond to the second scan, in experiments performed under conditions: scan rate of 60°C/hour, Pre-period 15 min, Gain high, Filter 10
The two profiles are identical: a pre-transition (from gel (Lb' or Lb) to rippled phase (Pb’)) and the main transition (in this case from rippled phase to liquid crystalline phase (Pb' to La)) are clearly observed.
Over more than 5 independent liposome preparations, it was found that there was no significant difference between the thermodynamic parameters retrieved as a result of the two preparation methods used for DMPC, within the reported margins of error.
The thermodynamic parameters retrieved from DSC analysis of liposomes prepared according to the two methods (where the subscript pre refers to the pre-transition, and m refers to the main transition) together with the uncertainty reported as twice the standard deviation of the mean, for 11 experiments (from the same and independent preparations taken together), using a scan rate of 60°C/hour, Pre-period 15 min, Gain high, Filter 10, are reported below:
Tpre = (15.4±0.4)°C ∆Hpre = (4.7±0.3) kJ mol-1
Tm = (24.1±0.1)°C ∆Hm = (27.0±0.7) kJ mol-1
Tpre = (14.7±0.4)°C ∆Hpre = (3.9±0.2) kJ mol-1
Tm = (24.4±0.2)°C ∆Hm = (26.2±0.6) kJ mol-1
MLVs and LUVs show quite different DSC profiles, as the peak for the gel to liquid crystalline transition is much more cooperative (i.e., much smaller width at half height, WHH), due to a larger number of lipid bilayers in the former ones.
In Figure 4 we can see the DSC traces for 3 mM samples of DMPC liposomes in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4), as MLVs (A) and LUVs (B), and the two curves superimposed for easier comparison (C).

Figure 4. DSC traces of MLVs (A) and LUVs (B) from DMPC in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4). Superimposition of MLV and LUV DSC traces (C). Liposomes (MLVs) were prepared by the film method. Experimental conditions were: scan rate of 60°C/hour, Pre-period 15 min, Gain high, Filter 10
To calculate the enthalpy of transition, we used the lipid content obtained by weighing for the MLVs and obtained from the phosphomolybdate method for the LUVs. The pre-transition usually either cannot be observed for the LUVs, or it appears convoluted with the main transition (as shown in Figure 3B). When the pre-transition and main transition appear convoluted, integration should be performed from the temperature at which the curve starts to deviate until it returns to the baseline, as the two peaks cannot be deconvoluted. In this case, we performed all LUVs integrations between 12°C-35°C, i.e., the main and pre-transition taken together.
The values obtained from 7-10 scans either from the same or different sample preparations (always using the film method), from DSC experiments using a scan rate of 60 oC/hour, Pre-period 15 min, Gain high, Filter 10 are shown below:
Tpre = (14.7±0.4)°C ∆Hpre = (3.9±0.2) kJ mol-1,
Tm = (24.4±0.2)°C, ∆Hm = (26.2±0.6) kJ mol-1
WHH = (0.6±0.1)°C
Tm = (24.6±0.1)°C ∆Hm = (26.8±0.3) kJ mol-1
WHH = (1.0±0.1)°C
In this set of data, we could not observe a significant difference between MLVs and LUVs in the temperatures obtained for the main transition (Tm). Regarding the values for the change in enthalpy, ∆Hm, the values reported do not precisely reflect the same phase change – in the case of MLVs, the values refer to the enthalpy change for the transition from rippled to liquid crystalline phase (Pb' to La) whereas for LUVs the reported DHm values reflect the enthalpy change for the transition from gel to liquid crystalline phases (see Figure 1).
Finally, the difference in WHH is very significant, as expected, since the value for LUVs is almost twice that retrieved for MLVs.
The results obtained agree with values reported in the literature[20,33] for similar studies. A wide range of values can be found, however, which stresses the importance of proper preparation and integration protocols, as well as the need for a full report of the experimental conditions.
Three different scanning rates were tested, for both MLVs and LUVs, namely 30°C/hour, 60°C/hour and 90°C/hour. The various scan rates were combined with the choice of filter to maintain a similar number of points in all cases.
| Scan Rate (°C/hr) | Filter | Tpre (°C) | ∆Hpre (kJ.mol-1) | WHH (°C) | Tm (°C) | ∆Hm (kJ.mol-1) | WHH (°C) | |
|---|---|---|---|---|---|---|---|---|
| MLV | 30 | 20 | 14.0 | 3.9 | 2.1 | 24.2 | 2.6 | 0.33 |
| 60 | 10 | 14.2 | 3.4 | 2.1 | 24.2 | 26.0 | 0.46 | |
| 90 | 8 | 14.2 | 3.5 | 2.1 | 24.1 | 26.1 | 0.40 | |
| LUV | 30 | 20 | - | - | - | 24.3 | 25.9 | 0.88 |
| 60 | 10 | - | - | - | 24.6 | 26.9 | 1.05 | |
| 90 | 8 | - | - | - | 24.6 | 25.8 | 1.08 |
As observed in the table, for both MLVs and LUVs neither the transition temperatures nor the transition enthalpies vary significantly with scan rate, within the observed overall uncertainty for the same liposome preparation (±0.3°C for temperature and ±0.5 kJ.mol-1 for enthalpy). Furthermore, even the WHH of the transition varies only moderately, which is understandable considering the high cooperativity of these lipid transitions, and justifies the use of high Gain for this type of experiment.
Although it is advisable to use a high Gain when working with liposomes, as previously mentioned, we tested the different Gain settings both for MLVs and LUVs. Results were collected for the same sample, scanned consecutively at different Gain settings.
In Figure 5 we plotted results for scans performed on MLVs (to see the effect on both the pre- and main transitions) with three Gain settings, the full curves (A) and an enlargement of the curve around the transition (B) where the differences in profile are clear. In the pre-transition, the differences are marginal, but in the main transition it is clear that the curve gets sharper and reaches higher specific heat capacity (Cp) values as the Gain increases, and the tail on the right increases as the Gain decreases.
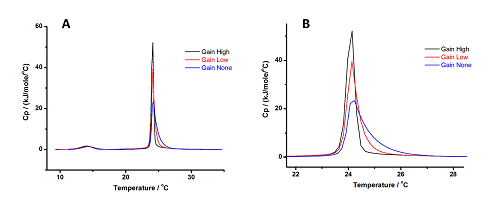
Figure 5: Results obtained for MLVs of DMPC at 3 Gain settings. Panel (A) shows the full curves, while Panel (B) shows an enlargement of the curve around the transition, for a clear view of the differences in profile at different Gain settings
The measured thermodynamic parameters are listed in Table 2.
| Gain | Tpre (°C) | ∆Hpre (kJ.mol-1) | WHH (°C) | Tm (°C) | ∆Hm (kJ.mol-1) | WHH (°C) |
|---|---|---|---|---|---|---|
| High | 14.1 | 3.6 | 2.1 | 24.1 | 26.0 | 0.3 |
| Low | 14.1 | 3.2 | 2.0 | 24.1 | 26.2 | 0.5 |
| None | 14.4 | 2.9 | 2.0 | 24.2 | 26.7 | 0.7 |
The enthalpy change of the pre-transition seems to decrease, whereas that for the main transition increases, as we move from High to No Gain. These effects are marginal, as overall the observed differences are within the combined uncertainties (± 0.3°C for the temperature and ± 0.5 kJ.mol-1 for the enthalpy change).
The effect of changing lipid concentration was tested to explore the detection limit of the equipment; i.e., to find the lowest possible concentration required for reliable results.
For this, a sample of MLVs was successively diluted 1:1, starting from a concentration of 3 mM and diluting down to ~20 mM, and each new solution was scanned under the same conditions. Results are shown in Table 3.
| Concentration (mM) | Tpre °C) | ∆Hpre (kJ.mol-1) | Tm (°C) | ∆Hm (kJ.mol-1) |
|---|---|---|---|---|
| 3.06 | 14.2 | 3.6 | 23.8 | 26.1 |
| 1.53 | 14.3 | 4.8 | 23.8 | 25.7 |
| 0.764 | 13.1 | 4.3 | 23.6 | 25.0 |
| 0.382 | 13.3 | 5.8 | 23.6 | 25.2 |
| 0.191 | 13.7 | 3.2 | 23.8 | 20.8 |
| 0.0955 | - | - | 23.8 | 17.1 |
| 0.0478 | - | - | 23.8 | 23.3 |
| 0.0239 | - | - | 23.8 | 19.1 |
Note: results are for the same lipid preparation, thus the uncertainty can be taken as (± 0.3°C for the temperature and ± 0.5 kJ.mol-1 for the enthalpy change).
It is remarkable that the temperature of the main transition can still be detected, and at the same value, for a concentration as low as ~20 mM. For MLVs, reliable temperature results can be measured for both transitions at concentrations down to ~0.2 mM, and reasonable enthalpy change values down to 0.4 mM. At lower concentrations, the curves are difficult to integrate accurately, and the values are consequently variable and unreliable.
It was observed that aging affects the retrieved parameters, as would be expected. The results obtained for MLVs and LUVs were consistent for samples measured up to 2 weeks after liposome preparation, but after this time the material does deteriorate. It is therefore not advisable to use samples more than 2 weeks old, even if kept in the refrigerator in dark containers to avoid oxidation.
Complementary experiments were performed with lipid mixtures, as well as for a lipid mixture (model membrane) and an antimicrobial peptide. DMPC and DMPG in a 3:1 molar ratio are presented as an example of a lipid mixture with close-to-ideal miscibility and behavior, and POPE and POPG, also in a 3:1 molar ratio, as a lipid mixture with pronounced non-ideal behavior. These two lipid mixtures have been used as model membranes to mimic pathogen membranes (in particular fungus) in antimicrobial peptide studies[20,26,37-39]. All liposomes were prepared in HEPES buffer (10 mM HEPES,150 mM NaCl, pH 7.4).
Liposomes were prepared by the lipid film method (lipid mixture), and then extruded according to the protocol described above. Due to the charge in the membrane (from the PG component) these lipid mixtures form oligolamellar vesicles (OLVs); i.e., before extrusion only a couple of bilayers exist in the liposomes.
The experiments were performed with OLVs at 60°C/hr, for the temperature range
10°C-35°C, Pre-period 15 min, Gain high and Filter 10. The results can be seen in Figure 6.
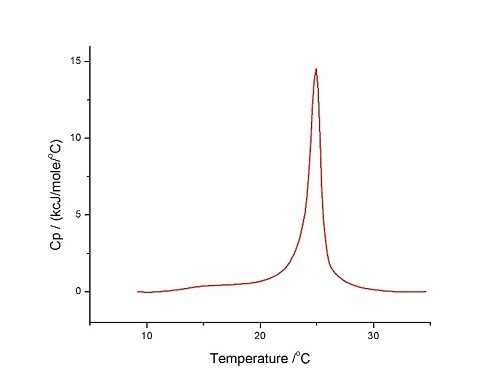
Figure 6. DSC analysis of LUVs of DMPC/DMPG 3:1 molar mixture in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4) performed at 60°C/hr, for the temperature range 10°C-35°C, Pre-period 15 min, Gain high and Filter 10
The curve is quite symmetrical and is similar to those reported above for pure DMPC LUVs, showing the close-to-ideal mixing for these two lipids. Knowing that the thermodynamic parameters for the pure lipids are:
DMPC
Tm = 24.6°C ∆Hm = 26.8 kJ mol-1 (this study)
DMPG
Tm = 23.7°C ∆Hm = 26 kJ mol-1[38]
It is clear that the mixture shows parameters similar to the independent components, as we obtained here (for different sample preparations):
Tm = 24.9°C±0.5°C ∆Hm = 27±3 kJ mol-1 (this study)
This lipid mixture is widely used in model membrane studies as a bacterial membrane mimetic. The two lipids mix non-ideally, showing non-symmetrical profiles particularly above xPOPG=0.5[40]. The parameters for the pure lipids are:
Tm = 24.7°C and ∆Hm = 20.5 kJ mol-1 for POPE, and:
Tm = -5.3°C and ∆Hm = 20.5 kJ mol-1 for POPG[40].
The analysis was also performed at 60°C/hr, for the temperature range 10°C – 35°C for OLVs and 4°C ‑ 35°C for LUVs; Pre-period 15 min, Gain high and Filter 10. Results are shown in Figure 7.
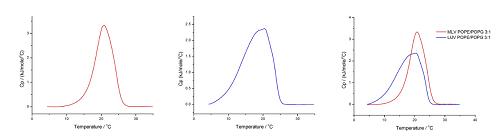
Figure 7. DSC analysis of POPE/POPG 3:1 in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4) performed at 60°C/hr, for the temperature range 10°C - 35°C for OLVs (A) and 4°C - 35°C for LUVs (B) (superimposition is shown in (C)), Pre-period 15 min, Gain high and Filter 10
Our analysis of the POPE/POPG 3:1 mixture shows a Tm of 20.7°C for OLVs and 20.4°C for LUVs, which is in agreement with the values obtained by other groups (for OLVs, 22.7°C for xPOPG=0.18 and 20.6°C for xPOPG=0.30[40], and 20.4°C for LUVs[41]). For the transition enthalpy, we obtained ΔtransH of 22 kJmol-1 for OLVs and 24 kJmol‑1 for LUVs, which is in agreement with the literature (24.2 kJmol-1 for xPOPG=0.18 and 25.5 kJmol-1 for xPOPG=0.30[40] and for LUVs, 22 kJmol-1[41]).
Model membranes are widely used in biophysical studies as they provide a simple system in which the properties can easily be changed in a systematic way to perform factor analysis; for example, changes of charge, pH, lipid chain length, saturation, lipid head group, buffer, ionic strength, and so on.
DSC provides an excellent first screening tool for the interaction of drugs, peptides and proteins with membranes of varying composition. Using the case of antimicrobial peptides (AMPs) as an example, their interaction with membranes mimicking eukaryotic cells and pathogenic membranes has been shown to be easily tested by DSC[20,21,26,42,43], with excellent correlation with microbiology studies[38,39].
CA(1-7)M(2-9) is a hybrid peptide containing amino acids 1-7 from cecropin A and 2-9 from melittin[44], which is known to be active against a wide variety of pathogenic agents[44-46]. Its interaction with membranes of varying composition has been studied by DSC, providing very interesting conclusions that correlate well with its antimicrobial action and hemolytic activity. To test the interaction parameters, the membrane charge should be changed to mimic either eukaryotic cell membranes or pathogenic cell membranes.
Inter-sample variation can impact the thermodynamic parameters measured by DSC analysis of different preparations of the same liposome suspension. Therefore, when performing these studies, the same batch preparation should be used for all peptide/lipid mixtures to be tested.
The pure lipid is run under the same conditions, and serves as the baseline for comparison of the behavior of the mixtures, as we are assessing the changes in lipid behavior induced by the presence of the peptide. In the case of AMP/membrane studies, the peptide should be added to the pre-prepared MLV or LUV suspension, in order to mimic antibiotic use. During analysis of the effects of the AMP, one should observe the profile changes (these can reveal membrane disruption or induced segregation), together with changes in Tm (stabilization/destabilization of gel phase; i.e., better/worse interaction in liquid crystalline phase) and ∆Hm (liposome disruption, relative importance of the peak components if segregation exists).
To illustrate this type of study, we show below (Figure 8) the changes in the DSC profile and temperatures for mixtures of POPE/POPG 3:1 LUVs with CA(1-7)M(2-9) at different molar ratios (P:L). It can be observed that at low peptide content, the main transition is sharper (more cooperative) and appears at a slightly higher temperature, but as the peptide content continues to increase, we observe membrane segregation (1:40) and significant destruction (1:15). The detailed discussion can be found in [41].
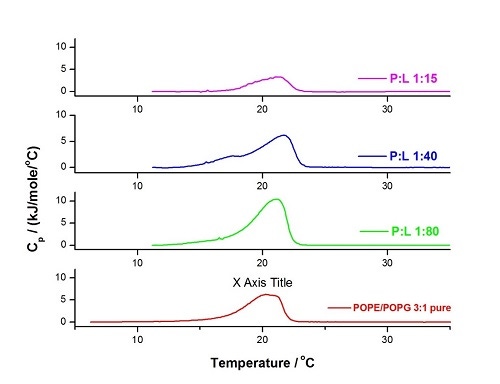
Figure 8. DSC experiment of LUVs of POPE/POPG 3:1 in HEPES buffer (10 mM HEPES, 150 mM NaCl, pH 7.4) performed at 60°C/hr, within the temperature range 5°C - 35°C, Pre-period 15 min, Gain high and Filter 10. Analysis was performed for the pure lipid mixture (red) and for 3 mixtures at different peptide to lipid molar ratios
DSC is a fundamental technique for the characterization of membrane behavior, providing all thermodynamic parameters for temperature-induced transitions. Furthermore, for mixtures of membranes with other components (drugs, proteins, peptides, polymers, etc.) DSC represents a first-line screening method, providing information on the effect(s) of the added components on membrane behavior.
The quality and reproducibility of the retrieved parameters depends critically on preparation and handling protocols, such as sample preparation, accurate concentration determination, experimental set-up, cleaning procedures. Furthermore, a clear statement of all details in subsequent reports is fundamental for the comparison of data obtained in different labs.
Thanks are due to strategic project Pest-C/QUI/UI0081/2011 and Pest-C/QUI/UI0081/2013 from Fundação para a Ciência e Tecnologia (FCT) and European Social Funds for CIQ-UP. MB would like to thank her students who completed DSC membrane work over the years: Filipa Abrunhosa, Regina Adão, Guangyue Bai, Vitor Teixeira, Tânia Silva, Inês Martins and Susana Correia. A.C.P. Águas is a F.C.T. doctoral fellow (SFRH/BD/88958/2012).
1. Chapman, D., Biological Membranes. 1968, London: Academic Press.
2. Goñi, F.M., The basic structure and dynamics of cell membranes: An update of the Singer–Nicolson model.BBA - Biomembranes, 2014. 1838(6):1467-1476.
3. Lindner, R. and H.Y. Naim, Domains in biological membranes.Exp. Cell Res. 2009. 315(17):2871-2878.
4. Nicolson, G.L., The Fluid-Mosaic Model of Membrane Structure: Still relevant to understanding the structure, function and dynamics of biological membranes after more than 40years. Biochim. Biophys. Acta, 2014. 1838(6):1451-1466.
5. Goñi, F.M., A. Alonso, L.A. Bagatolli, R.E. Brown, D. Marsh, M. Prieto, and J.L. Thewalt, Phase diagrams of lipid mixtures relevant to the study of membrane rafts. (BBA) – Mol. Cell Biol. Lip, 2008. 1781(11–12):665-684.
6. Marsh, D., Cholesterol-induced fluid membrane domains: a compendium of lipid-raft ternary phase diagrams. Biochim. Biophys. Acta, 2009. 1788(10):2114-23.
7. Vaz, W.L.C. and T.P. Begley, Lipid Bilayers: Properties, in Wiley Encyclopedia of Chemical Biology. 2007, John Wiley & Sons, Inc.
8. Mouritsen, O.G., Life - As a Matter of Fat: The Emerging Science of Lipidomics. 2005: Springer.
9. Liposome Technology: Interactions of Liposomes with the Biological Milieu. 3rd ed. Vol. III. 2006, New York: CRC Press.
10.Liposomes, Lipid Bilayers and Model Membranes. From Basic Research to Application. . 2014, Boca Raton: CRC Press, Taylor & Francis group.
11. Allen, T.M. and P.R. Cullis, Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev., 2013. 65(1):36-48.
12. Arouri, A., M. Dathe, and A. Blume, Peptide induced demixing in PG/PE lipid mixtures: A mechanism for the specificity of antimicrobial peptides towards bacterial membranes? BBA - Biomembranes, 2009. 1788:650–659.
13. Blume, A. and P. Garidel, Lipid Model Membranes and Biomembranes, in Handbook of Thermal Analysis and Calorimetry, pages 109-173. R.B. Kemp, Editor. 1999, Elsevier Press: Amsterdam.
14. El Maghraby, G.M., B.W. Barry, and A.C. Williams, Liposomes and skin: From drug delivery to model membranes. Eur. J. Pharm. Sci., 2008. 34(4–5):203-222.
15. Hancock , R.E.W. and A. Rozek, Role of membranes in the activities of antimicrobial cationic peptides.FEMS - Microbiology Letters, 2002. 206:143-149.
16. Matos, C., J.L.C. Lima, S. Reis, A. Lopes, and M. Bastos, Interaction of antiinflammatory drugs with EPC liposomes: Calorimetric study in a broad concentration range.Biophys. J., 2004. 86(2):946-954.
17. Pattni, B.S., V.V. Chupin, and V.P. Torchilin, New Developments in Liposomal Drug Delivery. Chem. Rev., 2015. 115(19):10938-10966.
18. Sandström, M.C., L.M. Ickenstein, L.D. Mayer, and K. Edwards, Effects of lipid segregation and lysolipid dissociation on drug release from thermosensitive liposomes.J. Control. Release, 2005. 107(1):131-142.
19. Van Vlierberghe, S., P. Dubruel, and E. Schacht, Biopolymer-Based Hydrogels As Scaffolds for Tissue Engineering Applications: A Review.Biomacromolecules, 2011. 12(5):1387-1408.
20. Abrunhosa, F., S. Faria, P. Gomes, I. Tomaz, J.C. Pessoa, D. Andreu, and M. Bastos, Interaction and lipid-induced conformation of two cecropin-melittin hybrid peptides depend on peptide and membrane composition. J. Phys. Chem. B 2005. 109(36):17311-9.
21. Arouri, A., M. Dathe, and A. Blume, The helical propensity of KLA amphipathic peptides enhances their binding to gel-state lipid membranes. Biophys. Chem., 2013. 180-181:10-21.
22. Bai, G., M. Nichifor, and M. Bastos, Association and Phase Behavior of Cholic Acid-Modified Dextran and Phosphatidylcholine Liposomes.J. Phys. Chem. Lett. 2010. 1(6):932-936.
23. Benesch, M.G.K. and R.N. McElhaney, A comparative calorimetric study of the effects of cholesterol and the plant sterols campesterol and brassicasterol on the thermotropic phase behavior of dipalmitoylphosphatidylcholine bilayer membranes.BBA - Biomembranes, 2014. 1838(7):1941-1949.
24. Blume, A., Applications of calorimetry to lipid model membranes, in Physical properties of biological membranes and their functional implications., pages 41-121. C. Hidalgo, Editor. 1988, Plenum Press: New York.
25. Ivanova, V.P., I.M. Makarov, T.E. Schäffer, and T. Heimburg, Analyzing heat capacity profiles of peptide-containing membranes: cluster formation of Gramicidin A. Biophys. J., 2003. 84:2427-2439.
26. Teixeira, V., M.J. Feio, and M. Bastos, Role of lipids in the interaction of antimicrobial peptides with membranes. Prog. Lipid Res., 2012. 51(2):149-177.
27. Chiu, M. and E. Prenner, Differential scanning calorimetry: an invaluable tool for a detailed thermodynamic characterization of macromolecules and their interactions.J. Pharm. Bioallied Sci., 2011. 3(1):39-59.
28. Garidel, P. and A. Blume, Miscibility of phospholipids with identical headgroups and acyl chain lengths differing by two methylene units: Effects of headgroup structure and headgroup charge. BBA - Biomembranes, 1998. 1371(1):83-95.
29. Garidel, P. and A. Blume, Miscibility of phosphatidylethanolamine-phosphatidylglycerol mixtures as a function of pH and acyl chain length.Eur. Biophys. J., 2000. 28(8):629-638.
30. Garidel, P., C. Johann, and A. Blume, The calculation of heat capacity curves and phase diagrams based on regular solution theory.J. Therm. Anal. Cal., 2005. 82(2):447-455.
31. Garidel, P., C. Johann, and A. Blume, Non-ideal mixing and fluid–fluid immiscibility in phosphatidic acid–phosphatidylethanolamine mixed bilayers.Eur. Biophys. J., 2011. 40(7):891-905.
32. Caffrey, M., LIPIDAT A Database of Thermo Data and Association Information on Lipid. 1993, Boca Raton Anne Arbor London Tokyo: CRC Press.
33. Koynova, R. and M. Caffrey, Phases and phase transitions of the phosphatidylcholines. Biochim. Biophys. Acta, 1998. 1376(1) 91-145.
34. Marsh, D., Structural and thermodynamic determinants of chain-melting transition temperatures for phospholipid and glycolipids membranes.BBA - Biomembranes, 2010. 1798(1):40-51.
35. Marsh, D., Handbook of lipid bilayers. 2nd edition. 2013, Boca Raton: CRC Press - Taylor & Francis Group.
36. McClare, C.W., An accurate and convenient organic phosphorus assay.Anal. Biochem., 1971. 39(2):527-30.
37. Bastos, M., T. Silva, V. Teixeira, K. Nazmi, Jan G.M. Bolscher, Sérgio S. Funari, and D. Uhríková, Lactoferrin-Derived Antimicrobial Peptide Induces a Micellar Cubic Phase in a Model Membrane System. Biophys. J., 2011. 101(3):L20-L22.
38. Bolscher, J.G.M., R. Adão, K. Nazmi, P.A.M. van den Keybus, W. van ’t Hof, A.V. Nieuw Amerongen, M. Bastos, and E.C.I. Veerman, Bactericidal activity of LFchimera is stronger and less sensitive to ionic strength than its constituent lactoferricin and lactoferrampin peptides. Biochimie, 2009. 91(1):123-132.
39. Silva, T., R. Adão, K. Nazmi, J.G.M. Bolscher, S.S. Funari, D. Uhríková, and M. Bastos, Structural diversity and mode of action on lipid membranes of three lactoferrin candidacidal peptides.BBA - Biomembranes, 2013. 1828(5):1329-1339.
40. Pozo Navas, B., K. Lohner, G. Deutsch, E. Sevcsik, K.A. Riske, R. Dimova, P. Garidel, and G. Pabst, Composition dependence of vesicle morphology and mixing properties in a bacterial model membrane system.BBA - Biomembranes, 2005. 1716(1):40-48.
41. Teixeira, V., M.J. Feio, L. Rivas, B.G. De la Torre, D. Andreu, A. Coutinho, and M. Bastos, Influence of Lysine Nε-Trimethylation and Lipid Composition on the Membrane Activity of the Cecropin A-Melittin Hybrid Peptide CA(1−7)M(2−9). J Phys Chem B, 2010. 114(49):16198-16208.
42. Epand, R.F., P.B. Savage, and R.M. Epand, Bacterial lipid composition and the antimicrobial efficacy of cationic steroid compounds (Ceragenins). BBA - Biomembranes, 2007. 1768:2500–2509.
43. Epand, R.M. and R.F. Epand, Domains in bacterial membranes and the action of antimicrobial agents. Mol. Biosyst., 2009. 5:580-587.
44. Andreu, D., J. Ubach, A. Boman, B. Wåhlin, D. Wade, R.B. Merrifield, and H.G. Boman, Shortened cecropin A-melittin hybrids Significant size reduction retains potent antibiotic activity.FEBS Lett., 1992. 296(2):190-194.
45. Chicharro, C., C. Granata, R. Lozano, D. Andreu, and L. Rivas, N-Terminal Fatty Acid Substitution Increases the Leishmanicidal Activity of CA(1-7)M(2-9), a Cecropin-Melittin Hybrid Peptide. Antimicrob. Agents Chemother, 2001. 45(9):2441-2449.
46. Rodríguez-Hernández, M.J., J. Saugar, F. Docobo-Pérez, B.G. de la Torre, M.E. Pachón-Ibáñez, A. García-Curiel, F. Fernández-Cuenca, D. Andreu, L. Rivas, and J. Pachón, Studies on the antimicrobial activity of cecropin A–melittin hybrid peptides in colistin-resistant clinical isolates of Acinetobacter baumannii.J. Antimicrob. Chemother, 2006. 58(1):95-100.