In some industries, for example, in early-stage pharmaceutical development, it is desirable to measure particle size and size distributions using as little as 5mg of dry powder material. In this application note, we demonstrate the capability of the Aero S dry dispersion unit and the Mastersizer 3000 to measure small quantities of pharmaceutical materials with good reproducibility, illustrating the dispersion abilities of the Aero S.
The routine measurement of particle size plays an important role in pharmaceutical and other industries, where it is often a critical-to-quality attribute for the product. Dispersion is critical for any particle size measurement and two principle routes are available in laser diffraction systems, wet and dry. If the powder is processed in a dry state and the end product is a dry powder, then a dry dispersion method is generally preferred. In the case of the pharmaceutical industry, active ingredients and excipients are generally handled dry during processing, and may be a dry powder in the final form for example in dry powder inhalers. In the early stages of product development, a pharmaceutical manufacturer may wish to measure the particle size of a sample of dry powder with less than 100mg of material in existence.
In this application note, we measure aliquots as small as 5mg of fine grade, pharmaceutical lactose and show that the measurement reproducibility is well-within the ISO laser diffraction standard [1] guidelines on repeatability. This fine grade of lactose was chosen as both an established model material for active pharmaceutical ingredients and as a highly cohesive powder that is difficult to disperse.
Aliquots of lactose were weighed out and handled with care to avoid powder compaction or adhesion to the weigh boat. An aliquot of 5mg was chosen as this allows for 20 measurements from the available 100mg, providing sufficient aliquots for both method development and reproducibility testing.
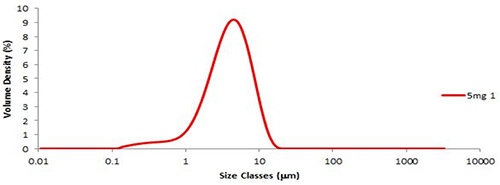
Six aliquots of the fine grade lactose were prepared and dispersed using the Aero S. A typical particle size distribution is shown in figure 1. The variation in the results, or reproducibility, was less than 0.8% for Dv (50) (table 1A). The ISO standard [1] states that the variation between repeated measurements (RSD%) should be less than 3% for Dv (50). As fine lactose is a particularly cohesive material achieving such low variation between measurements illustrates the excellent dispersion performance of the Aero S.
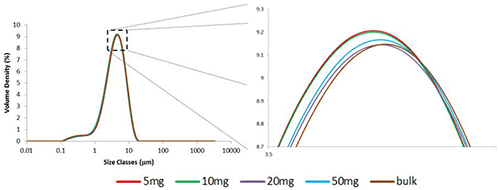
A mass titration was preformed to relate the 5mg aliquot to the bulk material and confirm that the 5mg result is typical. Three aliquots were measured of 5mg, 10mg, 20mg, 50mg and a bulk measurement of approximately one teaspoon. The particle size distributions are compared in figure 2. It is necessary to expand the graph significantly to separate the measurements. This indicates a very high level of consistency between the results. The variation between the measurements of aliquots of different mass, as quantified by the RSD, is less than 1.5% for all three percentiles (table 1B). This is well within the ISO standards [1] guidelines for repeat measurements of the same sample. To achieve RSDs well within the ISO guideline for repeatability from reproducibility data highlights the ability of the Aero S to disperse different masses of sample consistently.
|
| A | DV (10)(µm)
ISO 5% | DV (50)(µm)
ISO 3% | DV (90)(µm)
ISO 5% |
|---|---|---|---|
| 5mg 1 | 1.38 | 3.93 | 8.40 |
| 5mg 2 | 1.36 | 3.89 | 8.25 |
| 5mg 3 | 1.36 | 3.91 | 8.19 |
| 5mg 4 | 1.36 | 3.88 | 8.26 |
| 5mg 5 | 1.39 | 3.95 | 8.33 |
| 5mg 6 | 1.36 | 3.96 | 8.43 |
| 1RSD (%) | 0.949 | 0.795 | 1.11 |
| B | DV (10)(µm)
ISO 5% | DV (50)(µm)
ISO 3% | DV (90)(µm)
ISO 5% |
|---|---|---|---|
| 5mg | 1.38 | 3.93 | 8.40 |
| 10mg | 1.37 | 3.92 | 8.38 |
| 20mg | 1.38 | 3.99 | 8.54 |
| 50mg | 1.39 | 3.99 | 8.52 |
| Bulk | 1.41 | 4.06 | 8.61 |
| 1RSD (%) | 1.14 | 1.37 | 1.15 |
|
In addition to achieving a good dispersion, the reproducibility of results for small aliquots will be dependent on the median size and span of the distribution. If the median particle size is small, as it is here, then even 5mg will contain tens of thousands of particles. Assuming that the sample has been mixed homogenously, we can be confident that this 5mg sample will be representative of the whole population. As particle size increases, the number of particles in the sample will drop into the hundreds, and a 5mg sample may become less representative of the bulk. For example, if a coarse lactose with a Dv (50) of 100µm were measured in a 5mg aliquot, there would probably be higher variability in the percentiles reflecting the greater difficulty of sampling. Hence, it may not be appropriate to measure a coarse material in such a small aliquot. Similarly if the span of the distribution were large, indicating a polydisperse sample, the mass of the aliquot is limited by the ability to sample from the large end of the distribution.
To determine the mass required to make reproducible measurements, a mass titration similar to the one described above should be performed. The minimum aliquot will be the smallest mass with RSDs that satisfy the guidelines in the ISO standard [1].
Some industries, including pharmaceuticals, have a requirement to measure very small quantities of dry powders. We have measured a model pharmaceutical material in 5mg aliquots using the Aero S dry dispersion unit and the Mastersizer 3000. The results from 5mg were found to have variability of less than 1% RSD, indicating that the Aero S has excellent dispersion capabilities for fine powders. The results from the 5mg aliquot are in close agreement with those obtained from a bulk measurement.
[1] ISO 13320:2009 “Particle Size Analysis – Laser Diffraction Methods”.